Leeham News and Analysis
There's more to real news than a news release.
Bjorn’s Corner: Why e in ePlane shall stand for environment, Part 8. Hydrogen.
February 7, 2020, ©. Leeham News: After discussing established ways of improving the environmental footprint of our air transport system and highlighting the challenges involved with an electric/hybrid route we now look at hydrogen as an alternative energy source.
We will quote from a study series made by Airbus at the turn of the century. It’s today 20 years old but its ideas and conclusions are more relevant than ever.
Hydrogen as an energy source for airliners
Hydrogen has been a jet engine fuel since day one, its use predates kerosene. Hans von Ohain, the German designer of the first flying jet engine, used hydrogen as the fuel for his rig tests of the He-S-2 jet engines in 1937. It was subsequently found easier to use kerosene as it was easier to handle than hydrogen. But hydrogen is easily substituted for jet fuels in a gas turbine.
Hydrogen was considered as a replacement for kerosene at several points during the development of the jet engine, especially for high altitude military reconnaissance jets during the 1950s.
In the 1990s is was studied again by a team of Russian and German OEMs (Tupolev, Kuznetsov, and DASA, then an Airbus member). The found benefits and problems were spelled out in an Airbus presentation from 2001. The presentation is very clear and concrete and the study findings are still valid, despite the different studies being 20 to 30 years old:
Benefits:
- Hydrogen can be produced by electrolysis of water using any type of electrical energy.
- When hydrogen burns it produces water, closing the cycle.
- Hydrogen has 2.8 times higher energy density than jet fuel, meaning the carried fuel mass is reduced accordingly.
I would add to this: Hydrogen is also consumed during the flight, reducing the fuel mass, different to energy from batteries which have constant mass (presently 70 times heavier than jet fuel, data from a recent press release about Rolls Royce’s electric race plane).
Problems:
- Hydrogen in liquid form (LH2) has 4 times greater volume than jet fuel.
- It’s then stored in tanks at -253°C for reasons of volume and weight.
The lower fuel mass is a real plus. It enables long-range airliners as the fuel mass will be significantly reduced. An Airbus A330-300 crossing the Atlantic burns about 42 tonnes of jet fuel. When using LH2 this would reduce to 15 tonnes.
But the 4:1 volume ratio and low storage temperatures mean the design of the aircraft is influenced. The fuel would need tanks and pipes with good insulation and the tanks need to be spherical or cylindrical. This forces a different design of the aircraft.
Two studied concepts were presented in the Airbus presentation, one based on an Airbus A310 (Figure 1) and another based on a Dornier Do328 (Figure 2).
The A310 based aircraft was a result of a minimum change approach, Figure 3. The areas of the aircraft that needed change were analyzed in the study. They are listed on the graph from the presentation:
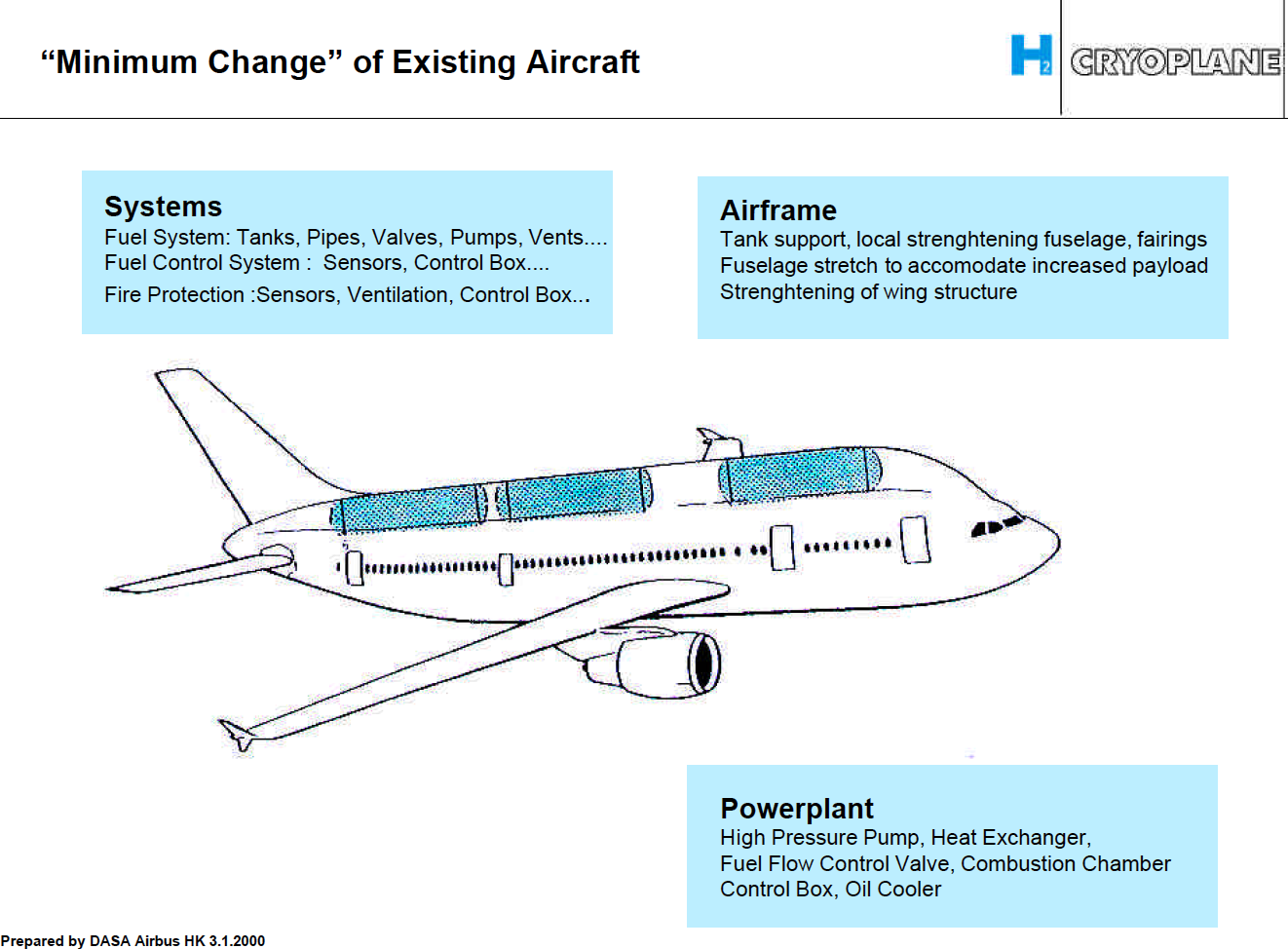
Figure 3. The A310 based “Cryoplane” with changes from a standard A310. Source: Airbus presentation.
The conclusions from the studies are 20 years old but interesting still today:
- Practical configurations could be made for all categories of airliners
- The Maximum Take-Off Weight of long-range airliners would reduce with 15%.
- Empty weight, on the other hand, increases with 20-25%.
- The energy consumption per seat-mile would increase by about 10%
The conclusion doesn’t list the most important advantage even though this is mentioned as a key goal in the initial part of the presentation, CO2 emissions from the engines when operating the aircraft would be 0%.
A hydrogen fuel ecosystem is missing
The Airbus/Russian industry studies had as one important goal lower CO2 emissions. In 2001, when the 10 years of studies with the Russian industry was summarized in this presentation, the pressing problem of CO2 wasn’t large enough to warrant further work along these lines. Today this has changed. The study goals and the results are highly relevant today.
When I look at the changes to what is a standard A310, the problem areas seem more manageable than the ones I see for electric/hybrid, for all airliners except the extreme short-haul ones. The study of the A310 variant had a minimum change approach. Given the storage requirements of LH2, a very different aircraft configuration like a Blended Wing Body (BWB) might be a better choice.
There are several problems associated with hydrogen-based aircraft. The present production of hydrogen uses natural gas, a fossil fuel. And the infrastructure to produce and distribute LH2 as aviation fuel is missing. This all needs further work and research.
Modern materials and better configurations could improve the performance data compared to the presented results. But the CO2 results need no improvement. They are game-changing.
My digging into the subject leads me to believe a longer-term solution to air transport’s environmental problems is hydrogen-based rather than electric/hybrid. We should direct our investments accordingly.
In next week’s Corner, we look at the alternative to burning hydrogen, use it in electric fuel cells.





Hydrogen as renewable, pollution neutral fuel has one major issue:
Losses in the circular process are unpleasantly high.
Hydrogen has some significant environmental negatives. It leaks at 4x the rate of natural gas (methane) through cracks. At the moment fugitive methane leakage rate is 1.2% in the USA and 0.6% in Australia so we could expect up to 4 times those typical rates. Furthermore fugitive hydrogen rises and destroys the ozone layer and seems to have 4 times the warming effect of C02 (due to its water creating tendency, CO2 is actually a poor greenhouse gas). Furthermore it eventually escapes to space depleting the planet of hydrogen/water. At an scale as large as the current petrochemical/gas infrastructure this technology is potentially dangerous. I suspect it should be confined to narrow industrial processes only such as iron smelting. It needs to be treated with caution. Hydrogen actually diffuses by dissolution through metals.
Methane is probably also an interesting candidate. CFRP tanks are being tightened with hardened epoxy and nanoparticles, so they can be quite light.
Not if you are looking at reduced carbon. Replacing one fossil fuel ( kerosene) with another, methane is pointless
Carbon neutral Methane can be made via methanation ie sabatier reaction.
https://en.wikipedia.org/wiki/Sabatier_reaction
https://matthey.com/en/products-and-services/chemical-processes/chemical-catalysts/methanation-catalysts
Of interest if CO2 can be obtained either from direct air capture or a concentrated source such as cement calcination. Methane is still lighter than air. There is a plant in Iceland producing 5000 litres per year of methanol from 10MW of geothermal power. It’s associated with a nearby Haber-Bosch ammonia plant. Catalysts are Copper Zink instead of Nickel.
Methane does not release as much water into the upper atmosphere as hydrogen would. It is easier to liquify and compress than hydrogen.
I suspect having gone to the trouble of extracting CO2 one may as well make a traditional fuel by using other catalysts though methane does not down stream refining issues.
you can burn all the carbon you want.
But your fuel must have been created from “fresh” carbon.
The issue with fossil fuels is that you release carbon into the atmosphere that has been sequestered over very long intervals many, many million years ago. you increase the carbon mass available to the environment.
Correction: 5000 tons (not litres) of methanol is produced in Iceland from CO2 extracted from air, hydrogen from electrolysis and geothermal power of about 10MW.
@William
It’s interesting to note that your ill-informed claims about a hydrogen economy were debunked 17 years ago by Amory B Lovins (Rocky Mountain Institute):
https://d231jw5ce53gcq.cloudfront.net/wp-content/uploads/2017/05/RMI_Document_Repository_Public-Reprts_E03-05_20HydrogenMyths.pdf
Let’s use the figures you provide of 0.04% escape to space. Current per capita oil consumption in a western nation is about 10L Per day, not including natural gas and coal. This is equivalent to about 3kg of hydrogen.
Assuming 3kg/day x 0.01 (leakage) x 0.0004 (space escape) x 7 x10e9 (billion people) we end up with atmospheric hydrogen escape of 84 tons/day ie about 720 tons of water. This doesn’t include hydrogen from incomplete combustion. They’re now injecting hydrogen into the gas network so simply leaving a stove burner unignited will lead to leakage.
Predominant Atmospheric escape of hydrogen on Earth is due to Jeans escape (~10 – 40%), charge exchange escape (~ 60 – 90%), and polar wind escape (~ 10 – 15%), currently losing about 3 kg/s of hydrogen.
https://en.wikipedia.org/wiki/Atmospheric_escape
A hydrogen economy will increase this by 33% if your sources references of only 0.04% escape to space are correct, which I wouldn’t trust without confirmation since as it’s a zealous lobby group.
And what of the effect on ozone? Hydrogen has a place but piping it to consumers is not one. It’s effects need to be studied more. PtL fuels represents a safer path.
@William
All estimates of the present rate of hydrogen escape from Earth are so small that the present water inventory could not be depleted by more than some 0.01% over the age of the planet.
The volume of all water on, in, and above the Earth — i.e. not including the water held in the Earth’s Mantle — is about 1,386,000,000 cubic kilometers.
With a water loss of 720 metric tonnes per day, all of the Earths water would only be depleted after a time span of 5300,000,000,000 years; or 5 thousand, three hundred billion years.
Bjorn – You haven’t looked at storing the hydrogen inside ammonia molecules. It doesn’t require cryogenic cooling, the pressure is far less (though you’d probably still need cylindrical tanks), you get more hydrogen per unit volume in ammonia molecules than simply compressing H2 molecules into liquid hydrogen, and there is a huge infrastructure for it already in existence – we produce a lot and transport it by road and rail for use on farms as fertilizer. You can burn it or you can put it into a fuel cell or both. It’s not as good as kerosene but it’s still a contender for relatively cheap, sustainably produced, carbon free fuel.
Unlike H2, the specific energy or NH3 is quite low. This effectively rules it out for aviation.
It’s still very good for terrestrial applications though, especially as converting it back to H2 absorbs heat from the surrounding air.
I wonder if Airbus studies also took into account the weight of all the necessary safety features to ensure the use of hydrogen with an acceptable level of risk.
In addition, since 2001, safety design requirements regarding explosionproofness have been dramatically tightened (for good reasons) and any new design has to cope with it (A350 included for example), with added weight.
Yes they did! This is the reason, why the hydrogen tanks are on top of the fuselage. Hydrogen is lighter than air and burns (only mixed 1:2 with oxygen it would explode) upward. You can evacuate an aircraft when it burns on top. This is opposite to liquid fuels, if they leak, the whole area around the aircraft might burn and could make an evacuation impossible.
Best example is the Hindenburg, where most passengers could be evacuated, while the hydrogen burned in a massive fire on top.
It’s worse than that I believe.There are two ways of producing hydrogen.One is from ‘cracking’ water which is clean but incredibly energy intensive ( breaking the chemical bonds).The other ( far more commonly used) is reforming methane or natural gas ,the left over CO is then mixed with water to extract further H2.
For every ton of H2 you get circa 9 tons of CO2……a problem!
That’s before you try to transport it and contain it economically.
Having said all that,oddly I am a supporter of H2 when used in ‘point to point’ situations such as aircraft and busses,as the storage and containment is far easier and controllable.But manufacture needs to be sorted first.
If Electrolysis infrastructure can be cheap and be economical to run with low capacity factor it could be perfect solution to utilize overproduction of renewable energy. We need to build renewable power with worst condition in mind, but then we get overcapacity in a good day. Just put it into hydrogen production, that is needed for aircraft or maybe even ships.
Some sites in Northen Germany do that, when they produce more wind power electricity than what can be transmitted to users they use the excess electrical power split water and pump the H2 into the natural gas network. Don’t know how efficinet catalysts they use but instead of wasting electricity.
They were using methanation ie combining it with CO2 over nickel catalyst to create synthetic natural gas but they may have switched to adding hydrogen gas directly. They’re planning to do this in the UK. I have misgivings in regards to the so called hydrogen economy. On a large scale, especially if delivered to the domestic consumer fugitive hydrogen has some considerable negatives to the environment.
Nevertheless it would be possible to set up small scale electrolysis, compression and liquefaction plants at an airport using the existing electrical supply network. This has been done at 2 german airports, on an experimental basis, to provide liquid hydrogen to cars and vans. The technology for cryogenic storage in cars has already been developed.
@William said: “I have misgivings in regards to the so called hydrogen economy. On a large scale, especially if delivered to the domestic consumer fugitive hydrogen has some considerable negatives to the environment.”
Another myth of yours debunked:
https://d231jw5ce53gcq.cloudfront.net/wp-content/uploads/2017/05/RMI_Document_Repository_Public-Reprts_E03-05_20HydrogenMyths.pdf
@OV-99. Lots and of money to be made in the new “green” hydrogen economy. Billions. Wind turbine manufactures have orders books with literally trillions lined up in both US and Europe. $ 50 billion being planed for hydrogen exports just from Australia.
Non of the claims you make are factually confirmed regarding fugitive hydrogen rates from either incomplete combustion or distribution leakage. Nor rates for atmospheric escape or ozone impacts. Before a headlong rush is made more is needed than the “Rocky Mountains Institute” doing a zealous series of arguments.
Domestic consumers are notoriously poor at maintenance.
It’s idiotic to rush into this.
@William
https://www.sciencedirect.com/science/article/pii/S0959378011000409
A company in Nebraska is using electricity to strip the C from methane to sell as carbon black, and then it sells the remaining hydrogen to produce electricity in a converted coal combustion plant. So that’s a cleaner way to make hydrogen than the usual reforming of natural gas.
https://journalstar.com/special-section/new-directions/monolith-investing-millions-in-nebraska-in-reinventing-the-carbon-black/article_a850a6d6-be35-5562-a482-2169c87c7e31.html
All you say about Ammonia is true and indeed well made points in terms of its far greater practical nature -except…
Once again ammonia is ( usually) made from natural gas and once again the by product is CO2.I believe about 4 tons of CO2 to one ton of Ammonia is the general figure for making NH2.I wish this wasn’t the case as Nitrogen by product from combustion hurts nobody.
Yes, I agree, you’d have to use renewable energy to hydrolise water to obtain hydrogen and then use more renewable energy and the Haber process (or perhaps a biological means) to combine it with atmospheric nitrogen. There’d be no point in it if manufacturing it if the waste product was CO2.
The CO2 can be easly captured and stored in ground and carbonize in some houndred years or so, like ccs.
So if the process is cheap and the gas is chesp its an interesting alternstive
It’s interesting that you mention ammonia. This is a key feedstock for critical fertiliser, explosives (mining) and chemical industry. If any industry that is to be decarbonised first it is ammonia production by Haber Bosch. Iron production will also decarbonise easily.
There have also been developments in hydrogen handling since those reports were written. For example, a recent approach that does not require subtantial (or any) insulation or strong pressure vessles and is much lower volume: http://www.research.lancs.ac.uk/portal/en/publications/-(2035473d-ee39-486f-8033-b056b87f3470).html
which is being taken forward by https://www.kubagen.co.uk. They are talking about cars but it does indicate that the storage issues landscape has changed.
At this point we do know that the energy density is very high for the demo product kubagen is promoting. So it would be amazing in cars and in land based transportation of hydrogen. What we don’t know at this point is the energy per weight, and if that is suitable for aviation. We also don’t know the ‘charge’ time, how long will it take to absorb the H2. Which is kind of critical. It’s a very promising material, though.
“Ohain, the German designer of the first flying jet engine, used hydrogen as the fuel for his rig tests of the He-S-2 jet engines in 1937. It was subsequently found easier to use kerosene as it was easier to handle than hydrogen.”
Anyone who has worked with hydrogen has found it maddeningly difficult to handle. It leaks. Infrastructure associated with it will experience losses. We might also ask where the hydrogen goes after it leaks out.
Yes, I agree, you’d have to use renewable energy to hydrolise water to obtain hydrogen and then use more renewable energy and the Haber process (or perhaps a biological means) to combine it with atmospheric nitrogen. There’d be no point in it if manufacturing it if the waste product was CO2.
Today’s edition of Offshorewind.biz contains the following article re Dutch research into storage of surplus wind energy. Hydrogen production mentioned as a route for this. Perhaps the aviation industry needs to start talks with the wind energy sector in order that ‘waste’ energy can be put to use?
https://www.offshorewind.biz/2020/02/06/dutch-research-council-awards-eur-1-1-million-for-offshore-wind-study/
Furthermore, bare in mind that fossil energy production can be carbon neutralised, using CCUS technology. There are energy costs to this, but the carbon can be stored or recycled. CCUS derived energy production used to produce hydrogen would be relatively CO2 free.
CCUS is currently being funded in the UK by government with a view to producing carbon neutral industrial hubs along the east coast in order to facilitate the reuse of redundant oil facilities for the reinjection of CO2 into the north sea (Acorn CCS, Pale Blue Dot Energy). Hydrogen production by such facilities from CCUS fossil energy (or surplus north sea wind generation) would be an ideal application.
Huge potential for H2 production?
The cost of capturing a kg of CO2 directly from the air is down to US$0.10 per kg. Papers being published suggest only 0.25 kWhr are needed to extract 1 kg from the air. One needs to capture about 3.1kg of C02 to compensate for 1kg of burned liquid hydrocarbon which produces 11kwHr of heat. The question then is what to do with it. One could geologically sequester it by pumping it deep under ground into various suitable formations for a further cost or one could covert it back to liquid fuels to create carbon neutral fuels. It will be a matter of cost. I can see that if one doesn’t purchase a carbon neutral fuel one will have to pay to sequester it or pay for a certified offset.
One thing is for sure, future aircraft will need to be very efficient because the 4 choices of airlines will be 1 a carbon neutral fuel such as hydrogen or PtL synthetic kerosene, 2 paying for CCUS sequestration, paying for traditional offsets or 4 electric.
German Government Estimates from 2016 estimated that a carbon neutral jet fuel could be produced from $1.5-$1.7 per liter. I think the price will easily drop 33% but but such prices will keep air travel as affordable as it had been if fuel burn per passenger in aviation is nearly halved.
CCUS May be cheaper for a a few decades than carbon neutral fuels but in the long run I’m sure we will run out of suitable places to sequester and be forced to turn to carbon neutral fuels.
One of the big factors in avaition’s contibution to climate change is the “high altitude non-CO2” emissions, these do nearly as much damage as the CO2 emissions.
Do we know if hydrogen would be the same / more / less? It makes a big difference as to whether hydrogen is a viable alternative (and in my opinion should be taken into account when considering all “green” alternatives).
This is a non technical article – there’s a little chart just before 1/2 way. But it gives an idea and uses UK government published data.
https://www.bbc.co.uk/news/business-51364103
Sorry if this is a spoiler for a future part in the series – and thanks for doing this Bjorn – it really is appreciated.
You will only get noticeable non-CO2 exhaust, like NOx, if you burn hydrogen hot. Unfortunately, the efficiency of a jet engine increases with the burning temperature.
This would not occur if you use ‘cold burning’ of hydrogen in a fuel-cell. But this would increase weight and complexity. I’m not aware of any viable alternative, to the fuel-cell powered hydrogen airplane, to eliminate NOx exhaust.
Using the cryogenic cooling capacity of hydrogen could be used to separate out the nitrogen. This was used in the Martin Marietta LACE “Liquid Air Cycle Engine” an air breathing rocket for a single stage to orbit vehicle. Exhaust gas recirculation would Reduce Nitrogen. Adding a super critical carbon dioxide closed cycle exhaust gas system to recover energy would improved efficiency and lower fuel burn. Micro-channel heat exchangers are getting very small. Super critical C02 turbines are 7 times smaller than steam turbines.
Just a note, the nox has been traced by radioisotope analysis to be coming from fertiliser use, not aviation, cars or diesels. An issue yes but also a degree of noise and hype.
Lots of research on cathalysts to reduce the energy required for hydrolysis, Stanford U is one that reports some success. Hydrogen means requirements on materials that can be brittle from hydrogen diffusion into the material, High Stength Steels is one of them.
If you burn hydrogen+oxygen in combustion you have a very quick burning and hot flame and I don’t know how much NOX is produced. Using fuel cells you aviod NOX and get higher efficiency than a gas turbine combustor+turbine but the Power per lb is limited, so it might get into a mix of wing mounted gas turbines and a APU/aft mounted boundary layer fan driven by fuel cells. The Cold LH2 could be used to cool the Engine compressor air increasing its effeiciency and become H2 gas before it gets into the burner.
One issue with hydrogen production is its efficiency. Electrolysis followed by liquefaction is about 57% efficient due to the large costs of liquefaction. That is a lot of bird and bat killing wind turbines and bird frying solar concentrator plants need to be built.
Granted the latent heat of liquefaction can be used to operate cryogenic or superconduct equipment on an aircraft but I suspect it can be used to precool the air of an engine core to just above the pinch point of air as it is in the SABRE engine thereby reducing compressor size. (Factor of 4 in sabre).
My own belief is that a technology called PtL “Power to Liquids” that creates a carbon neutral synthetic hydrocarbon jet fuel. It involves extracting C02 from the atmosphere, so called DAC “direct air capture” and combining it hydrogen obtained from electrolysis. This process is ready.
It is already happening: In Iceland the George Olah CO2 to Renewable Methanol Plant, Reykjanes is producing 5000 tons of methanol per year since 2012 using geothermal power that could be converted to 3000 tons of jet fuel via ZSM5 catalyst. Nordic Blue Crude AS will build the worlds first commercial Power-to-Liquid jetfuel plant at Herøya, Porsgrunn, Norway. 20MW of hydro power will produce 8000 tons of jet fuel per year. There are other plants in the world including US.
Concentrating solar power does not fry any birds. Check the research on aviary mortality due to concentrated solar power by Cliff Ho at Sandia Labs.
PV panels do not concentrate sunlight so no frying possible even if you wanted to (except eggs which fry just fint on the top surface a good day).
https://youtu.be/ICLXQN_lURk
There is more footage if you care to look.
Wind turbines are also destructive to raptors which are attracted to the motion and wind. I’ve heard of a bat kill of a million and a bird kill of half that in the US alone. Possibly an exaggeration but it’s certainly happening.
Individual concentrator dishes could possibly be enclosed in glass or mesh.
The industry is now worth trillions and expect it to put out “public relations”
There are trails with “flying” windmills that operate at higher altitudes with stronger winds. They are thus quiter, less visible, above most living flying birds and land when the wind slows down. The drawback are they are not as powerful yet and they require more advanced control systems.
A problem that never seems to be considered is the weight of the tether. Copper cable weighs a lot but is needed to get the power to the ground. https://wattsupwiththat.com/2012/09/10/enough-wind-to-power-global-energy-demand-except-when-the-wind-doesnt-blow/
Burning hydrogen in turbofan engines is a big waste of its potential I think.
Using hydrogen fuel cells to power electric propulsion systems is a way more efficient use for it.
Show us how that works without adding weight ?
Liquid Hydrogen… Think Space Shuttle Challenger explosion.
A plane full of liquid hydrogen would be a bomb-in-waiting. A slight “mishap” on landing for example could end up in a huge fireball.
This was my thought as well, wondering if the H on the first aircraft illustration stood for Hindenburg.
The cause of Hindenburg’s demise is unclear:
https://en.wikipedia.org/wiki/Hindenburg_disaster
The cause is immaterial. The fact that it occurred ended the use of hydrogen in airships.
It helped that helium was a better choice and available .
Its still a mystery why long distance airships didnt survive longer as the Hindenburg took 2 1/2 days to cross the Atlantic ( from Germany) and the fastest liners took 5. ( Southhampton to NY) More normal passenger ships could be 10 days.
Germany could not get Helium.
The US could get their airships to fly reliably. ( all crashed ?)
WWII started.
After the war airplanes had ascended to that role.
flying boat museum in Foines, Ireland has
a newspaper cutting exhibited tagged “the future of passenger flight” ( FW200 landing around New York coming non stop from Berlin Staaken)
FW200 may have gone non stop for that flight, but it was with a cabin full of fuel. More normal range with passengers would be Berlin to Athens. I was thinking of a transition period and the war of course sped technology of long distance winged flight up 10 fold..but for military purposes.
It was discovered that the Challanger crew survived the explosion and that the impact of the crew compartment on water killed them.
The fireball of burning hydrogen is impressive but far less dangerous than burning fuel on the ground around and below a fuselage.
Yes, terrifying but true:
http://www.nbcnews.com/id/3078062/ns/technology_and_science-space/t/chapter-eternity-descent/#.Xj8b2DFKj4Y
The shuttle had a range of rediclouse compromises. No ejection seats so that more crew could be carried, certainly no Mach 3.3 capable ejection seats, the solid rocket boosters an inappropriate technology (a kerosene/LOX booster with fly back capability similar to Buran would have been safer) too many disposable boosters to allow it to carry 30 tin keyhole satellites rather than scientific loads.
Elon Musk, a trained electrical engineer and a private entrepreneur , put the aerospace establishment industrial military complex to shame. He had a tiny fraction of their resources. A fly back booster was possible from the 1950s onwards.
A blow torch jet of super hot flame burning into a kerosene or LOX tank would have been as destructive as a hydrogen tank.
https://www.nationalgeographic.com.au/space/5-myths-of-challenger-shuttle-disaster-debunked.aspx
The explosion technically did not destroy the orbiter, but it immediately lost attitude control and breakup was very quick, within no more than 2 seconds after failure of the external tank. Only pieces emerged from the fireball.
The engines had shut down before telemetry ended and before breakup, due to high temperatures in the turbopumps. Also the shuttle experienced 0.2g’s of lateral motion in both directions from off-axis thrust, so was probably off-attitude already.
The shuttle was then deflected to the side by ignition of RCS fuel by the fireball. This attitude change imposed a load of 20g, four times higher than the 5g rating. The crew cabin separated and the shuttle bay exploded from the inside by aerodynamic pressure, leaving the engine/tail section intact but burning from RCS fuel.
Even if the shuttle had emerged unscathed, there was no abort or landing option for that altitude.
@BernardP
The myth presented by you that liquid hydrogen supposedly would be a bomb-in-waiting, has repeatedly been debunked over the years.
Here’s Amory B Lovins refuting your ill-informed claim:
https://d231jw5ce53gcq.cloudfront.net/wp-content/uploads/2017/05/RMI_Document_Repository_Public-Reprts_E03-05_20HydrogenMyths.pdf
I worked with hydrogen gas as a fuel source for an experimental thruster. It did require extensive protections in the form of gas and flame detectors, all interlocked with the fuel flow and possible sources of ignition. Also the test space had to be atmospherically isolated from the personnel space. These are safety concerns that did not exist with liquid fuels, but might equally apply to any gaseous fuel. So safety concerns are present but not insurmountable.
It would be better to use propane/butane mixtures. The energy content is similar to natural gas, but it can be stored at 8 bar. It is a waste product of the oil refineries. The most commonly used way of producing H2 from natural gas by reforming also releases CO2, electrolysis is far too inefficient, and the electricity production is not free of Co2.
These gases are heavier than air and will “pool” in the bottom of boats, houses and obviously aircraft rather than float away and disperse. Much more dangerous than methane. Also the end game is complete or decarbonisation of aviation. Synthetic Carbon neutral fuel is possible to make but propane/butane from fossile fuel won’t work.
Bjorn, why do the tanks need to be spherical? Better surface area to volume ratio? Or did you mean cylindrical?
Cooling/insulating at varying ambient conditions seems like the biggest challenge to me. Done with a low enough weight penalty, there’s probably a good case to be made for use with mid-range missions.
Thanks Albert, should be spherical or cylindrical. Fixed.
If you drive an FCEV in California now and fill up at one of the stations that store Liquid H2, you’ll find that some of that Liquid H2 came all the way from Quebec, Canada by truck. So if you can put Liquid H2 in a tanker truck for the several days it takes to make that journey from Quebec to California, I doubt it’s much of a challenge with insulation on an airplane that gets filled up right before departure. Just sayin’…
BMW had a vacuum walled tank for its 7 series car that used a modified piston engine about 20 years ago, it also had 30 layers of foil to reflect out infrared. The tank could store liquid hydrogen for 4 days, during which time the tank would build up pressure. After that the hydrogen was vented but consumed by a catalytic device. A 10-15 minute drive before the 4 days was up would consume the pressure build up. I believe they succeeded in a better tank that used magnets to suspend itself at several points and could be odd shaped. That tank could last 7-11 days before venting.
An aircraft would likely used “foam” or possibly a metallic sheet with an airgap could be developed.
The German navy uses metal hydrides, (lithium and titanium) to store hydrogen in solution on its Type 214 U-boats which have a PEM fuel cell based air independent propulsion. Great care must be taken that no oxygen contaminates the hydrides as it will poison the hydride. All sorts of methods need to be used. These would be suitable for smaller aircraft eg general aviation.
General Aviation will go Hydrogen Fuel Cell Electric with Compressed only 700 bar storage just like Hydrogen Fuel Cell cars do today. When it’s compressed and not liquid there’s no burn off, and you can store compressed gaseous Hydrogen much longer than you can store gasoline or diesel. Compressed hydrogen doesn’t degrade like fossil fuels do.
Got it, thanks Bjorn.
FlyInPeace, but on an airplane LH2 has to be fed to the engines as well, so its more complex than just a tank.
Yes, but Bjorn’s article overdramatizes this a bit imho. The typical timeframe between a commercial airliner gets refilled and the fuel is used poses no particular issues in terms of Liquid Hydrogen burn off. And if you do get some burn off you can use it for a Fuel Cell APU or for the engines. Not sure why pipes have to be insulated as the H2 has probably become gaseous at that point. The pipes in my FCEV are not insulated.
Oh interesting, but is transporting gaseous H2 “long” distances in an aircraft (depending on the tank location) safe without insulation? The gas pressure would need to be well controlled, right?
Typically you won’t be transporting gaseous H2 ‘long’ distances in an airplane. If it’s a ‘long’ distance the H2 will be liquid. If it’s a short(er) distance in a smaller airplane the H2 might be gaseous. There’s some tank insulation for gaseous H2 tanks, but it’s limited as the gaseous tanks are designed to handle over-pressure. For FCEVs there’s a filling standard based on outside temperature so that you never get over-pressure, even if you fill your tank in freezing temps and then drive your FCEV directly into a heated garage. For an airplane that’s not an issue since you fill up where it’s hot (the ground) and go up where it’s cold (the air). So the gaseous H2 will shrink and the pressure will go down as you fly up. Also: By the time you’ve done a take off you’ve already used up enough H2 to avoid any issues with temperatures and over-pressure for gaseous H2 tanks. It’s worth noting that airliners today handle 5,000psi hydraulics (A350 & A380 & more) in bending wings, so it’s obviously not that much of a leap to handle 10,000psi in a cylindrical tank. But most commercial airliners won’t handle compressed gaseous H2, they will use Liquid H2.
Spherical tanks are the lightest for a given volume, but space considerations dictate cylindrical tanks.
Bjorn,
Nice article. However, the major issue with hydrogen is that it is an energy carrier and not a not a naturally occurring energy source (like petroleum) to be just mined and used. It has to be produced, liquified, stored and transported and all these add to the overall carbon footprint, unless the energy to do all this is derived from the sun or wind (even those have carbon footprints due to manufacturing and other issues). Hydrogen embrittlement and potential leakage are major problems. Finally, as mentioned in these blogs, the safety issue may be a major certification hurdle. The increased empty mass also does not help. Given all these issues, the technology is nowhere near practical utility for the foreseeable future, imho, even though it will cut out CO2 emissions by aircraft. But then, aircraft are a very minor source of overall carbon emissions (less than 10% for the foreseeable future) and people like Greta are badly misinformed. We should be talking about major emitters like cars, trucks, power plants rather than beating up on air travel, which has connected the world in ways only internet and GNSS can beat!
Kant
What you say about hydrogen is true. It needs to be produced etc. But jet fuel does not just flow from the ground at airports. It needs to be discovered, extracted from deep under ground, transported vast distances, refined, transported again and stored. All that has its own CO2 footprint. Plus there is the issue of un-needed gases burned off as flares or just allowed to escape.
Hi Kant,
I drive an FCEV in California. The ’embrittlement problems’ you describe do not exist in the real world. It only exist on hydrogen hating blogs. The steel tanks used for low pressure storage at the Hydrogen stations are typically age tested for 50 year design life. Some high pressure tanks are certified for 20 year design life. Just FYI.
The intelligence of this community is refreshing! Every comment well considered. I do love Leeham News!
Here is a note on ‘electrolysis’ for water splitting into oxygen and hydrogen.
There are many ways of doing this but I will cover 3.
The traditional way is the alkaline electrolyser where a pair of electrodes are separated by a glass fibre mesh or asbestos and immersed in a solution of potassium hydroxide. By passing an electric current through hydrogen comes out of one side and oxygen out of the other. Efficiency is about 55% but can get to 75% but 55%-60% is more typical. The gas can be pressurised. Some people claim 85% by operating at elevated temperatures and using a steam turbine to recover energy.
This type of electrolysers was used in Norway, Canada and Egypt from the 1930s or earlier using cheap hydro power to make hydrogen that was combined with nitrogen to make ammonia and ammonium nitrate fertilisers and chemicals.
PEM Polymer Electrolyte Membrane Electrolysis uses a plastic membrane to separate a platinum and iridium electrode that is 80%-85% efficient. Unlike Alkaline cells they operate well with part load.
SOEC Solid Oxide Electrolyser Cell operate at between 500C-850C and can be from 85% to 95% efficient. They can potentially combine water and CO2 to make methane.
PEM and SOEC can operate in reverse as fuel cells.
SOFC solid oxide fuel cells can probably achieve 65% efficiency and if turbo-compounded could achieve 85% efficiency.
There are other ways: use a catalyst such as cerium oxide and hot steam from a High Temperature Reactor to split water directly.
Apart from using hydrogen directly it could be combined with CO2 to synthesis traditional hydrocarbon fuels, to make ammonia or to replace coke or methane in iron production. Incidentally Iron production is 5% of emissions.
http://www.psmsl.org shows actual se level rise, rate has not accelerated significantly, tectonic plate rise and fall is a larger effect.
http://www.thenewatlantis.com/archive/15/zubrin.htm the hydrogen hoaz
Yes, hydrogen is a way of transporting energy, not a source of it. However there is more than enough free energy available from the sun, and plenty of deserts that can be covered in PV cells (which are getting cheaper by the minute) to produce green energy, which that can then be used to produce cheap green hydrogen at the coast. Hydrogen has been used in industrial processes for many years and the technology for handling it is very mature. Recent resarch has found new catalysts for electrolysis, and new ways to store the energy (e.g. dissolved in ammonia, or stored in fibreglass tanks for cars that are safer than existing petrol tanks.) A plane with filled hydrogen tanks would be safer than one filled with kerosene as the hydrogen will disperse upwards. The real problem is the NOx produced by burning hydrogen in a gas turbine, which is the only way to use it while obtaining the same power to weight ratio as kero. A fuel cell and electric motors will be nowhere near as efficient.
Nikola is reporting that their cost of green H2, dispensed, is $3/kg using NEL Electrolyzer and dispenser, and Arizona green energy.
https://twitter.com/nikolatrevor/status/1220406200333824000?s=20
Maybe the next FSA from Airbus could be a step forward ?
If their MAVERIC demonstrator is an indicator of their current thinking, their next FSA might be 3 or 4 aisles. A 20% reduction of CO2 would be a step in the right direction.
“The airframer also says it is leaning on fly-by-wire know-how developed for combat aircraft to control the intrinsically unstable flying-wings.” – https://www.flightglobal.com/singapore-air-show-2020/airbus-studies-blended-wing-airliner-designs-to-slash-fuel-burn/136662.article
“intrinsically unstable” might be a bit challenging to get past the regulators though.
“It’s more likely to be the successor of smaller aircraft than a very big one,” says Dumont. – same article.