Leeham News and Analysis
There's more to real news than a news release.
Bjorn’s Corner: The challenges of Hydrogen. Part 4. Hydrogen safety.
August 14, 2020, ©. Leeham News: In our series on hydrogen as an energy store for airliners we are closing in on the design problems for a hydrogen-fueled airliner.
One of the aspects we must understand before discussion aircraft solutions are the safety aspects of hydrogen as a fuel in aircraft. Another is the requirements for the storage of liquid hydrogen, LH2.
The safety of hydrogen as a fuel for aircraft
When one thinks about hydrogen-fueled aircraft, the airship Hindenburg catching fire when landing at the Lakehurst Naval Station, NJ, comes to mind, Figure 1. The immediate reaction is: it seems dangerous, which is the wrong impression.
Hydrogen as a fuel has been used over the last 100 years for many vehicle types. One example is the space launcher industry. Others, where its use is growing, are cars, buses, trucks, forklifts, etc. For each area, there were extensive safety studies and tests before its use.
The elevator version of the results is:
It’s not more dangerous than hydrocarbon fuels like car gasoline or Jet-A1.
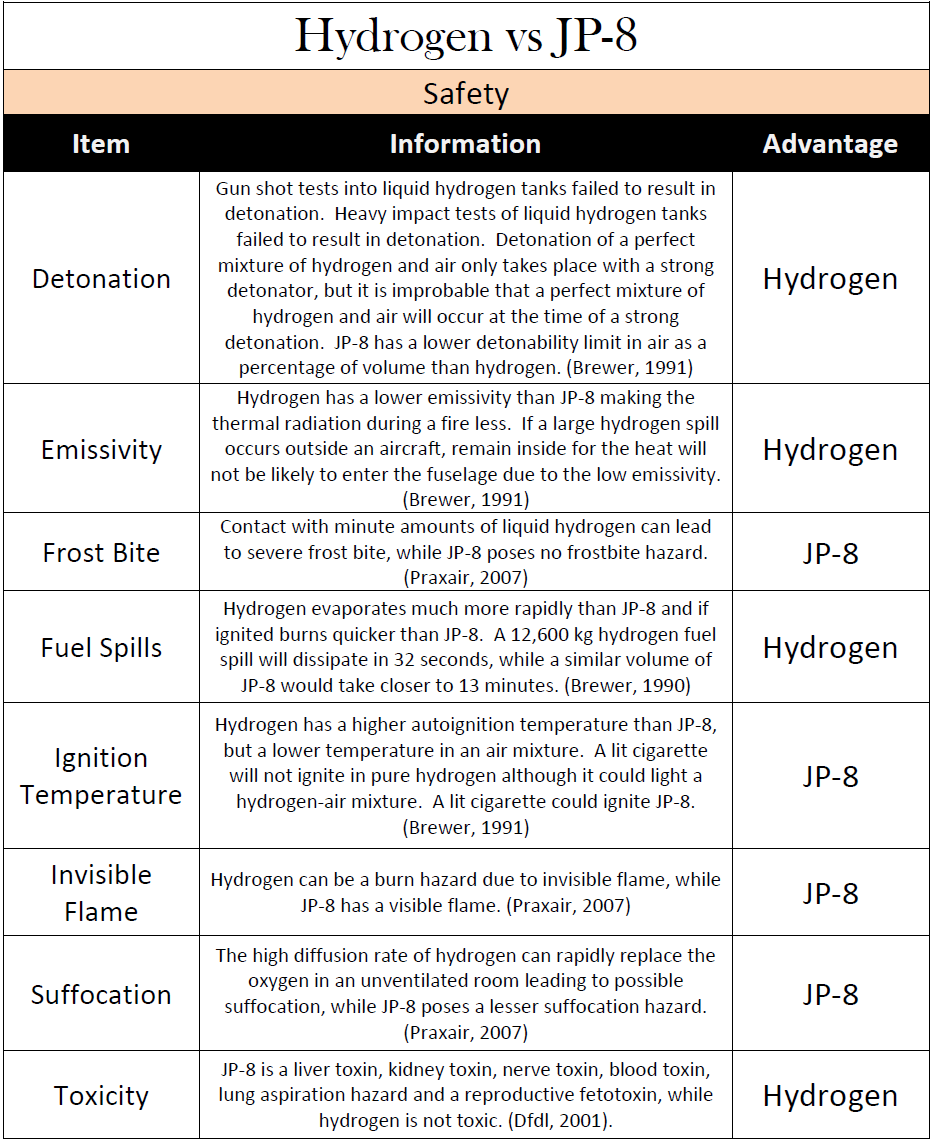
Figure 2 is from a good 2009 graduate research project by Adam D. Reiman at the US Air Force Institute Of Technology. He’s studied LH2 as an alternative to the US Air Force’s JP8 fuel (Jet-A1 with corrosion and anti-ice additives) for it’s Air Mobility Command.
His literature study examined all relevant work around hydrogen-fueled vehicles up to the reporting year of 2009. The report contains several tables comparing the fuels’ different aspects, where Figure 2 is the one comparing the safety aspects of JP8 to hydrogen.
Airbus’ Cryoplane study from 2001 comes to the same conclusions, Figure 3.
The bad image of hydrogen safety comes from the Hindenburg crash. The reality is that it’s in several aspects better than today’s jet fuels. It has characteristics that must be considered, but this applies to all fuels.
A leak will quickly evaporate, hence any leakage when fueling will benefit from the fast evaporation in free air, where H2 is a non-toxic substance. Leakage in closed compartments like tank and fuel system areas need detectors and ventilation safeguards.
The detonation risk is lower than with Jet-A1 and a fire burns quickly, with less heat than for Jet fuel. The fire is invisible, however, which is a problem.
Cryogenic tanks
We have learned we need LH2 (liquid hydrogen) to keep the storage volume down for airliners. This requires cryogenic tanks, where the LH2 is held at -253°C. This means double wall tanks, spherical or cylindrical, with isolation between the walls. We only need about one-third of the Jet-A1 fuel weight but it will take four times as much volume as this weight of Jet-A1 would take.
Jet kerosene as fuel has the advantage to use the wing structure as tanks (carefully sealed), which means it’s placed close to the center of lift and gravity, and the structure holding it is almost for free. For hydrogen, this will be difficult as it requires cylindrical tanks, and these will have a too large diameter to store in the wings (dividing the hydrogen over many tanks is not efficient).
Most studies have the tanks in the fuselage, above or aft of the passenger compartment.

Figure 4. Airbus Cryoplane study with tanks above the cabin for the A310 and on the wing for Do 328. Source: Airbus.
In Figure 4 an Airbus A310 has the tanks above the cabin (from the 2001 Cryoplane study). The study also looked at a regional Do 328 where the tanks were placed on the wings.





Thanks Bjorn. Do you see the LH2 tanks being cryo-pressurized, vented, or regulated at/near 1 atmosphere? If they are vented or regulated, then the possibilities for shapes may be extended. In that case the limiting factor would be heat transfer rather than structural strength. But heat transfer is also minimized for spherical or cylindrical tanks, so perhaps they remain preferred.
Hydrogen can cause Hydrogen embrittlement in alloy steels and put demands on all steel parts in the fuel system. Burning hydrogen stochiometric in air gives a high temperature of around 2100C, still GE stationary turbines has been hydrogren mixed in fuel up to 80% for years after they adapted the Engines and accessories for it. So careful engineering solves the Engine part and I assume the NOX as well. Putting in double walled LH2 tanks and tubing into an Aircraft of A320 pax capacity is a challange and it might result in a A322 size with conformal body tanks in the whole aft body and the fly-by-wire cables can be snugly fitted around the tank. This might open for new dry carbon wings on the A320H protoype.
Airports around EU will need decades to install LH2 tanks and processing Equipment for fueling/defueling and LH2 production, so both Air Liquid and Linde gas besides Areva and Simenes will have plenty of work for 50-100 years if the H2 passenger Aircrafts becomes real, eventually GE-Praxair will be competing for Airport work as well.
@claes
As for hydrogen embrittlement:
–
Page 17:
https://www.dlr.de/content/en/downloads/2018/university-of-stuttgart-polaris-future-aircraft-design-concept_3098.pdf?__blob=publicationFile&v=10
–
Page 6
https://www.dglr.de/publikationen/2018/480344.pdf
NASA is using friction stirr welded Al-Li alloy tanks for their Space Launch system tanks, so it might come as well as on the hydrogen Airbus and maybe on the rest of the fuselage as it is a bit stonger and corrods slower than regular aircraft aluminium alloys. The trend is anyway Al-Li in the fuselage and carbon fiber wings (777-9)
I would not be comfortable flying with cryogenic fuel over my head. A leak could be fatal. The fuel pods under the wings looks better, from a structural point also.
A naive question I’m sure, but could a switch be made whereby bags and freight could be carried in the front/middle of the wings, (rather like an overhead locker arrangement) and fuel tanks in the hold?
The issue would be safety in having a cryogenic fluid stored in the crush zone beneath the aircraft. Upon rupture, the cryo-fluid itself would be a hazard, and would also flash to lighter-than-air vapor, which if ignited would form a vertical jet of flame.
That’s why the designs are focused on storage either above the passengers, which has the cryo-fluid spillage problem but not the flame problem, or a fuselage extension at the rear which has some protection from both events.
The LH2 could be stored in separate twin engine booms for the greatest safety, but this would increase the wetted surface area & drag, which reduces the efficiency of the aircraft overall.
It may be that as tank engineering progresses, the tank shapes could evolve and be restored to the wings, but the required volume might necessitate something akin to a blended wing design.
@Rob
In DLR’s Gondola Fuselage Concept (link 2 below) the pressurised passenger cabin is separate from the unpressurised cargo compartment — i.e. with the cargo loaded into pressurised containers.
In the Polaris Concept (link 1 below) the LH2 tanks are located in the unpressurised cargo compartment underneath the pressurised passenger cabin.
The Gondola cargo compartment also serves as an energy absorbing element in the case of loading due to a crash landing and can be supplemented by additional crash absorbers.
Also, the lower panel of the pressurised passenger cabin — with approximately twice the radius of the side panels — can be outfitted with a sufficient flame retardent material that would withstand a vertical jet of a hydrogen combustion flame for some time.
–
https://www.dlr.de/content/en/downloads/2018/university-of-stuttgart-polaris-future-aircraft-design-concept_3098.pdf?__blob=publicationFile&v=10
–
https://www.dlr.de/fa/Portaldata/17/Resources/dokumente/institut/2003/2003_07.pdf
Yes, we discussed these in the last Corner. I doubt the lower hold storage concepts could ever be certified, for safety reasons. They are not mentioned in any of the Airbus concepts or the EU white paper.
As Bjorn mentioned in the series on electric aircraft, you can propose any number of arrangements and technologies, but having them certified to aviation standards is another matter.
Also the Boeing CHEETA concept for hydrogen powered aircraft has a blended wing design with hydrogen storage in the extended body/wings. So none of the manufacturer concepts have lower hold LH2 storage. The reasons have to do with safety.
And it is not even clear that the the H2 was even the cause of the Hindenburg Disaster…
https://en.wikipedia.org/wiki/Hindenburg_disaster#Incendiary_paint_hypothesis
@JD Evora
Twenty Hydrogen Myths
by AMORY B. LOVINS,
With respect to the Hindenburg disaster:
https://d231jw5ce53gcq.cloudfront.net/wp-content/uploads/2017/05/RMI_Document_Repository_Public-Reprts_E03-05_20HydrogenMyths.pdf
Semantics … most people who die ‘in fires’ are killed first by smoke and toxic fumes, similar to plane fires.
The burning of the gas in the Hindenburg was the factor which then led to the deaths.
Same way that lithium batteries are only the starting point in a plane fire, still makes them very dangerous.
@Dukeofurl
The combustion temperature of a lithium ion battery undergoes three stages: initial growth, sharp rise and decay. The difference between a benign outcome and a potential disaster is the necessity to take measures to extinguish fire and explosion in the initial growth stage – a hard thing to do on an aircraft in-flight.
Now, in spite of the vast quantities of hydrogen that was used as a lifting gas for the Hindenburg, no one in the accident was seriously injured or burned to death from hydrogen, which rapidly dissipated up and away from the passengers. This is in contrast to conventional hydrocarbon fuels where the hydrogen is chemically bonded to relatively heavy carbon atoms, which causes the fuel stick to people like glue and literally burn their skin off. Thus, the real lesson from the Hindenburg — a rigid airship built without fire retardant materials — is how safe hydrogen is if an accident is going to occur.
Duke is right about this. The contribution of hydrogen in the Hindenburg fire was the speed in which the structure was consumed. That required a far larger amount of energy than was contained in the doped skin or frame alone. Combustion of the hydrogen provided the needed energy.
The spectral emissivity of the flames was due largely to combustion of the skin and other materials. But evidence of hydrogen combustion is visible in the photo of the jet of flame emerging from the nose. There was a central walkway which provided a path for the burning hydrogen when pitched nose-up.
It’s true that an uncontrolled skin and/or frame fire would cause a crash of any airship, regardless of buoyant gas used. But estimates have shown a much longer period required if the gas was helium, on the order an hour or more, instead of 30 to 40 seconds as occurred wit the Hindenburg.
What sort of volume of LH2 would be required and how much would the pressure vessel containing it weigh?
In the EU hydrogen white paper Bjorn is using as a guide for this series, the combined weight of fuel & storage system is given as a gravimetric index (percentage of empty tank & systems weight to full LH2 tank). This is assuming that lightweight carbon-fiber tanks, which are the best available at present, can be improved somewhat over the next few years.
For a narrow-body shorter-range (2000 km) aircraft, the assumed gravimetric index value is 35%, which gives a total weight of 4 to 5 tons shifted from wings to the fuselage, divided between two tank systems.
The volume ratio would be about 3 to 1 over jet fuel for LH2 (plus storage), so roughly 14,000 gallons, 1900 cubic feet, 55 cubic meters. For a reference A321 with a 4 meter fuselage, this would add a fuselage section of about 4 to 5 meters in length. These are all rough estimates, to give an approximate idea of size and weight.
Hello Bjorn,
Can you please elaborate, in one of the next Corners, why LH2 tanks must be spherical or cylindrical. I understand this for pressured H2, but why can’t liquid H2 be stored in tanks of any shape.
Which materials are suitable as tank material?
KRJörg
LH2 tanks can be vented to atmosphere (with higher losses) or cryo-pressurized (with lower losses). The losses are due to heat transfer into the tanks, which evaporates the LH2.
Vacuum-bottle type insulation gives the lowest heat transfer. However it also depends on the ratio of tank surface area to contained volume of liquid. So spherical and cylindrical tanks have the greatest strength for cryo-pressurization, but also have the minimum ratio of surface area to volume, so lowest losses.
Lastly if double-wall vacuum insulation is used (as is likely), fabrication is easier for the simpler spherical and cylindrical shapes. In theory a vented tank of any shape could be constructed, but would be subject to the costs as outlined above
A risk is that leaks of hydrogen gas enter the cabin – the article speaks of the need for detectors (which are widely available and inexpensive) and ventilation. Would the risk of a combustible hydrogen/air mixture in the cabin lead to the requirement that the cabin shall be treated as a “hazardous location” requiring all electrical equipment to be intrinsically safe or explosion-proof?
If so, that would add dramatically to the cost and complexity and may mean the end of passengers powering up their electronic devices onboard.
Not sure about aircraft, but when working with LH2 in buildings, detectors and alarms were required, together with automatic shut-off and/or ventilation. But not explosion-proof fixtures.
The detectors are sensitive enough to pick up trace hydrogen well before the explosive or flammability limits. The white paper says that intra-aircraft transport systems for LH2 have yet to be developed. My guess is all systems and piping would be enclosed in externally vented containment at lower pressure, sealed from the passenger compartment at higher pressure, such that any venting or leakage would flow outside the aircraft. But we’ll have to see what develops.
No cabin lights, no food heaters, no…………………..
Hand held devices are not an open flash issue.
Hydrogen is clearly no different than propane or natural gas storage.
A bit tougher tech wise as you need the right materials but that is not new.
The Hindenburgh disaster will never be fully understood (sans building scale model and testing all the theories).
Natural gas sort of solved the problem with Macadam, houses still blow up.
Cars catch on fire as well.
Realistically Hydo issue goes back to tanks and unless fuel is taxed to hell and back, its non competitive.
Best route is to clean up jet engines and Jet A and concentrate (pun intended) on reducing ground based emissions to compensate.
In surveying, if your measurement of angle and or distance are off, you can find the error or you can fudge it to make it work. When you fudge it, its called a forced closure (illegal but its been done more than once)
Hydrogen as a fuel for Jet aircraft is a forced closure. The dog just don’t hunt.
Looking at history of fighters aircraft designs with more range you get to the de Havilland Mosquito and Lookheed Lightning with twin booms. Taking this concept to passenger aircrafts and hydrogen you get the twin A320 where one cabin or “pod” is the LH2 tank and the other carries pax. It migt be a quick and dirty design but with an A320neo pax load in one fuselage and LH2 in the other with the 35k neo engines soon being certified it is one step on the way. Most iarports can pretty easy take the increased span and the wings can be new slender dry carbon wings.
Clearly, public psychology (mentioned in one of the studies) plays a very important role in these matters. While favorable arguments can be made by comparison with jet fuel, the lay public may or may not appreciate them.
Two excellent examples: 1. The current situation with the corona virus. In spite of all the measures that have been taken to mitigate the risk, a significant majority of the public still refuses to fly (air traffic has recovered to only 30% of the pre-pandemic level and is stuck there. 2. The upcoming experiment with B737-MAX. We will see how many chose to fly in that aircraft, once it starts flying sometime this year, at least in the USA. My unsubstantiated guess would be something similar to the virus situation.
Unless the percentage of the public scared to fly is kept to single digits, the aircraft will not be economically viable (assuming the need for at least 80% load factor to break even). Hydogen-fueled aircraft, even if technically feasible, may not be economically viable. It will be hard to persuade an airframer to take that risk.
You start mixing up your data and you come up with bad results.
Even if air travel is 100% guarantee safe, you have the same issues of infection (de-boarding crowding is huge) and why do you travel? To see things, meet people, eat out all of which involved groups which in turn you have zero control over how they are doing.
And what people say vs what they do (MAX) in a fly in it or not fly are two different hings.
People spout a lot but when push comes to shove they cave.
Will a few refuse, sure. Enough to make any difference, no.
6 months down the air corridor no one will know the difference.